Odyssey: Pharmaceutical
Track radioactive materials, waste, as well as equipment related to production and testing of pharmaceuticals.
ODYSSEY WORKFLOWS FOR PHARMACEUTICAL
Cradle-to-Grave Inventory Management
Manage radioactive sealed and unsealed sources, leak testing, authorized limits, isotopes, users, locations, waste containers with automatic decay calculations, and custom compliance thresholds in accordance with your permit(s) or license(s). Includes a QR code labeling tool and a mobile-friendly interface to streamline access to information you need when you need it.
Asset Management
Comprehensive management of physical assets including machines, PPE, and portable equipment. Schedule and track inspections, surveys, and audits with fully customizable frequencies. Includes QR code labeling tools for efficient asset tracking and quick access to dedicated asset profile.
Analytical Reporting
Access to a centralized library of pre-configured reports designed to satisfy internal as well as regulatory requirements. Reports are fully automated, customizable, brand-ready, and can be saved as presets to ensure consistent and efficient reporting on a pre-determined basis.
Internal Workforce Process Enforcement
Manage inspections, surveys, safety data sheets, audits, assessments, questionnaires, and documented processes in one place. Submit processes for signature or approval, store them in dedicated profiles, and compile them for analysis and reporting.
Compliance Calendar
A centralized calendar for all due dates across Machines, Equipment, Site, and Personnel Inspections, Surveys, Audits, Training, Leak Tests, and more. In addition, it integrates with Outlook and Google, can be filtered to show only relevant events, and also supports scalable, role-based view permissions for every user.
Dosimetry Management
Direct integrations with Mirion, Landauer, and Radetco, plus advanced management tools including dashboards for abnormal readings, ALARA thresholds, and badge distribution tracking. Moreover, built-in response documentation allows you to record as well as manage investigations for anomalous dose readings.
Training
Create new courses or upload existing courses (PDF, PowerPoint, Video) for assignments, testing, and completion tracking. Furthermore, certificates are generated upon a passing grade, and reassignments can be scheduled on a recurring basis.
Incident Management
Record and manage hazards, injuries, illnesses, safety events, as well as near misses using structured case workflows. In addition, assign personnel and locations, request and collect information, and then monitor progress throughout the investigation process.
recommended modules to maximize workflows
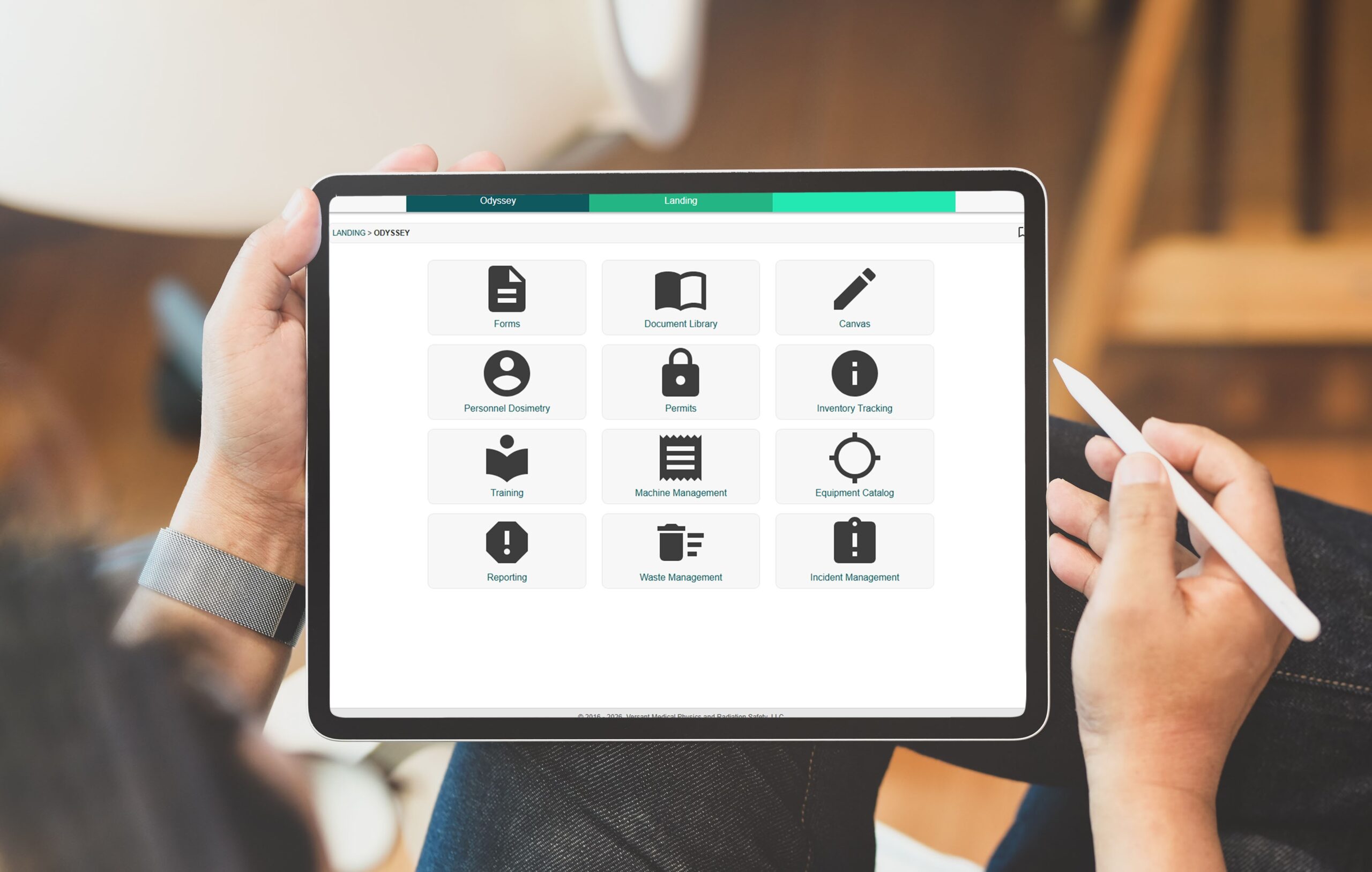
Inventory Tracking, Waste Management, Equipment Catalog, Machine Management, Personnel Dosimetry, Permits, Training, Reporting, Forms.
THE CHALLENGE
Like its healthcare counterpart, the pharmaceutical industry is both fast paced and critically important for the continued health and wellbeing of the public. As a result, it’s imperative that RSOs and EHS managers remain proactive in their approach to maintaining regulatory and environmental compliance. This is necessary to mitigate risk and provide continued safety for researchers, workers, and the patients using their products.
If you’re drowning in manual data entry and retrieval or constantly chasing down due dates, it can be difficult to focus on productivity and innovations that will make a difference in the lives of those you serve.

THE SOLUTION
Odyssey’s centralized, cloud-based platform streamlines your overall workflow processes and provides a simple way to manage your health and safety program. By taking the place of outdated spreadsheets and paper, Odyssey saves you time and money. Additionally, its system of checks and balances helps you ensure all necessary compliance tasks are completed and up to date. Ultimately, this allows you to focus on big picture opportunities in the marketplace.
